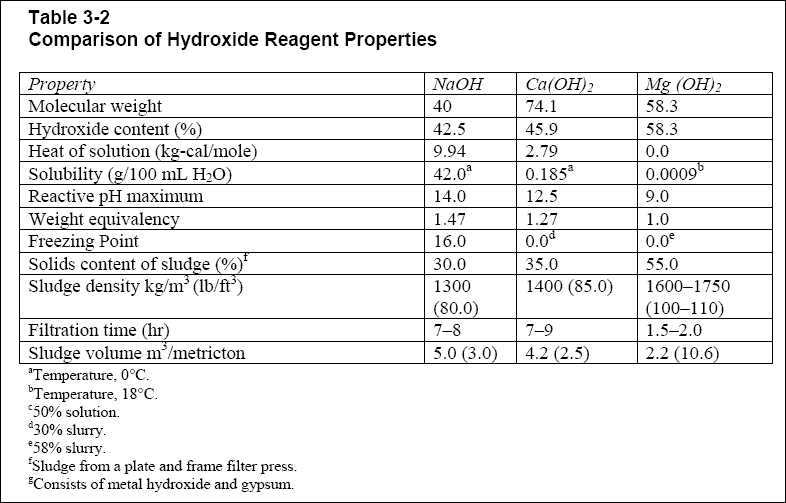
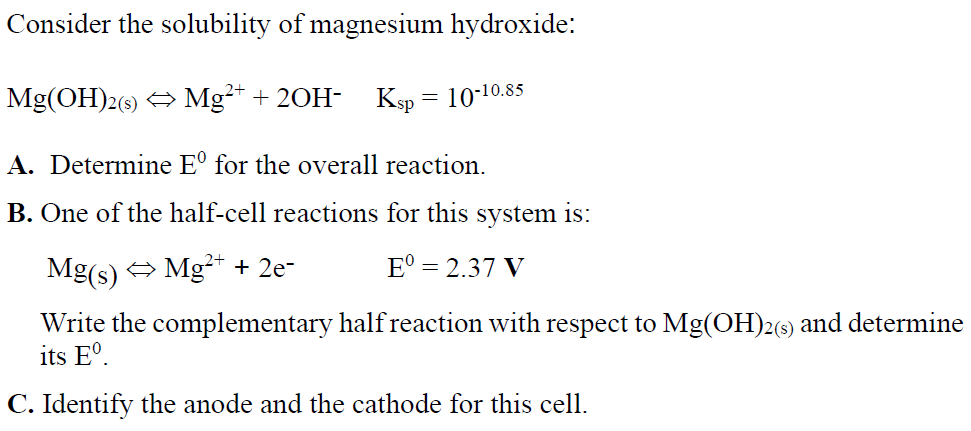pH of a saturated solution fo magnesium hydroxide in water at `298K` is 10. The solubility - YouTube

SOLVED: Question 66 0 / 1 pts What is the solubility of magnesium hydroxide; Mg(OH)z, if the solubility product is 2.00 x 10-11 (mol/L)3? t Answer 1.71X 10-4 mol/L 3.16 X 10-6

If the solubility of Mg(OH)2 in water is 1.4 × 10^-x . Ksp = 1.2 × 10^-11 . Find out the value of x.

Inhibition of Homogeneous Formation of Magnesium Hydroxide by Low-Molar-Mass Poly(acrylic acid) with Different End-Groups | Industrial & Engineering Chemistry Research
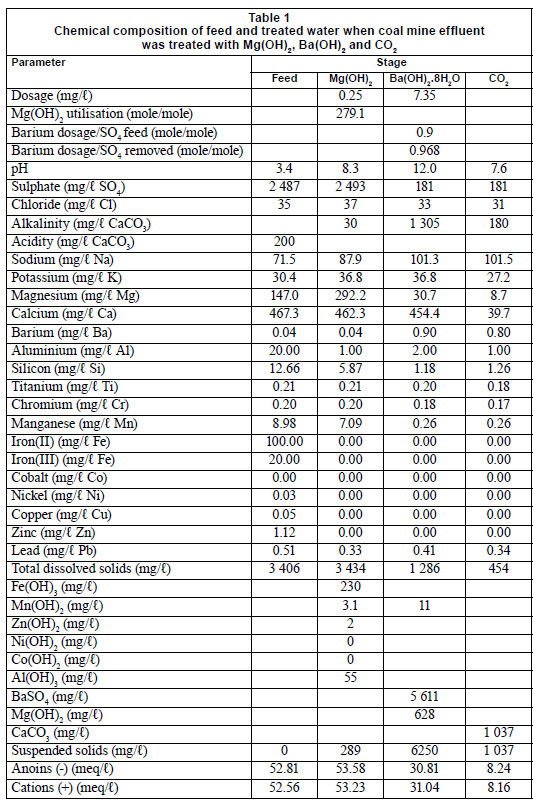
Application of magnesium hydroxide and barium hydroxide for the removal of metals and sulphate from mine water

Crystallization of calcium carbonate and magnesium hydroxide in the heat exchangers of once-through Multistage Flash (MSF-OT) desalination process - ScienceDirect
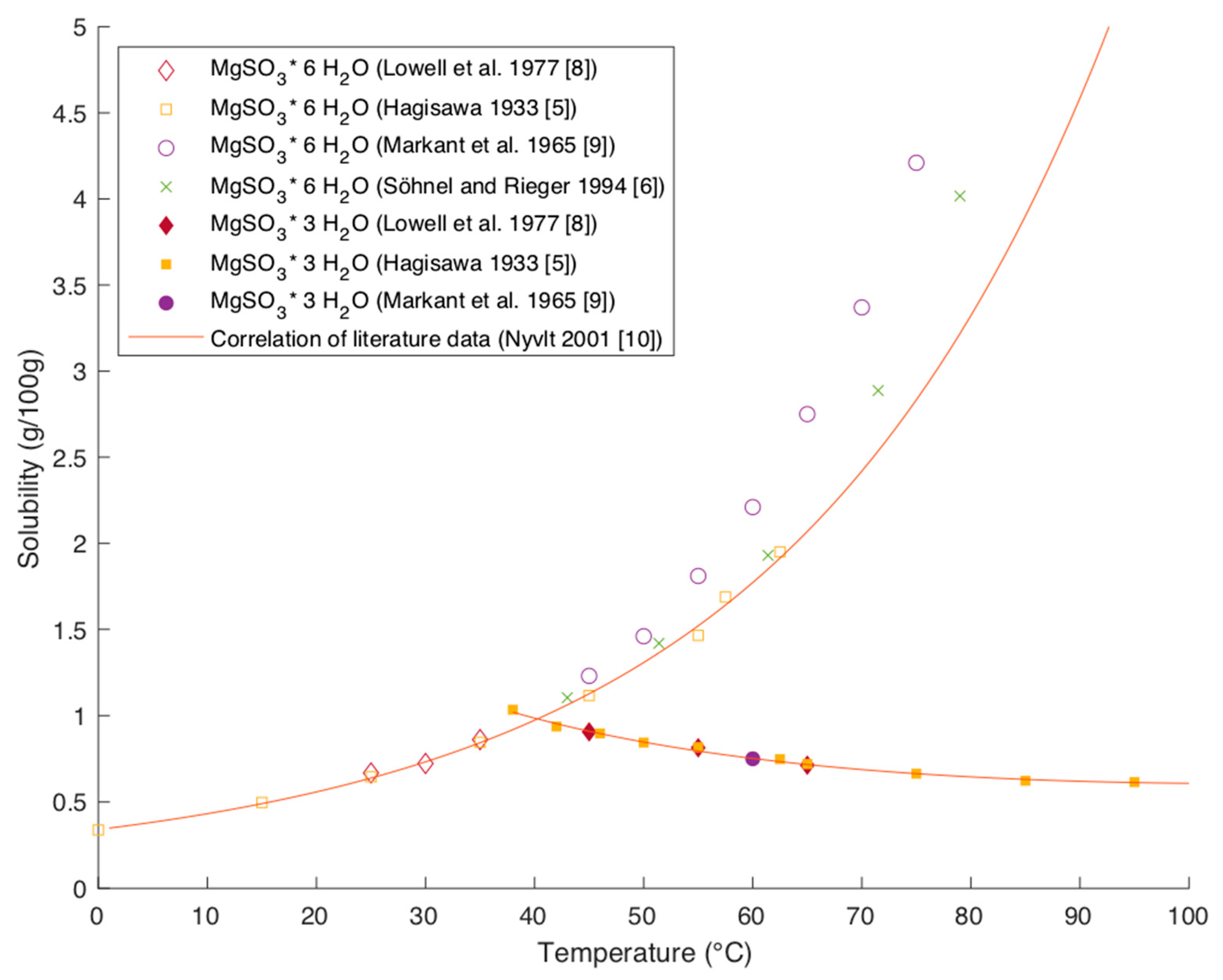
Processes | Free Full-Text | Solubility Data of Potential Salts in the MgO-CaO-SO2-H2O-O2 System for Process Modeling
![SOLVED: Calculate the solubility constant for dissolving Magnesium hydroxide Mg(OH) in water if [Mgl is 0.014 Problem 3: Calculate the solubility constant for dissolving Calcium hydroxide Ca(OH)z in water if [Ca ] SOLVED: Calculate the solubility constant for dissolving Magnesium hydroxide Mg(OH) in water if [Mgl is 0.014 Problem 3: Calculate the solubility constant for dissolving Calcium hydroxide Ca(OH)z in water if [Ca ]](https://cdn.numerade.com/ask_images/e1aaca9121da4955a58de8b427babda4.jpg)
SOLVED: Calculate the solubility constant for dissolving Magnesium hydroxide Mg(OH) in water if [Mgl is 0.014 Problem 3: Calculate the solubility constant for dissolving Calcium hydroxide Ca(OH)z in water if [Ca ]

The pH of a solution of B (OH)2 is 10.6 . Calculate the solubility and solubility product of hydroxide.