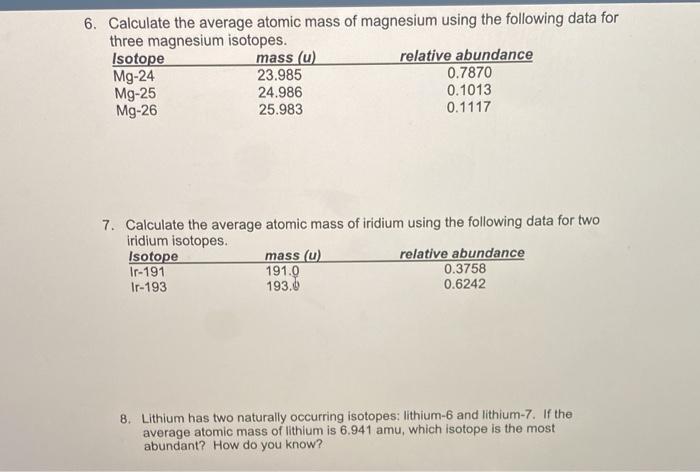
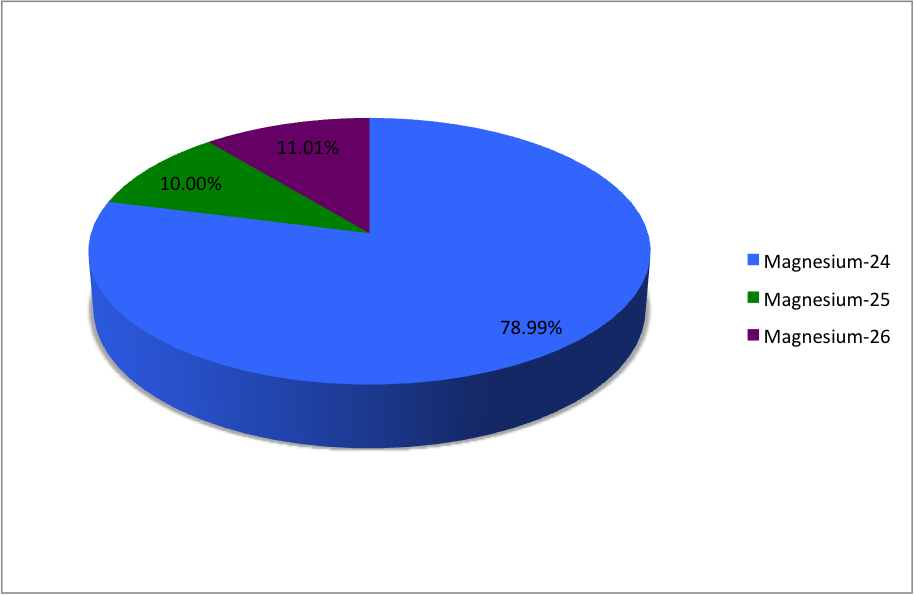
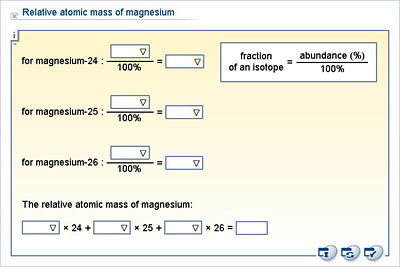
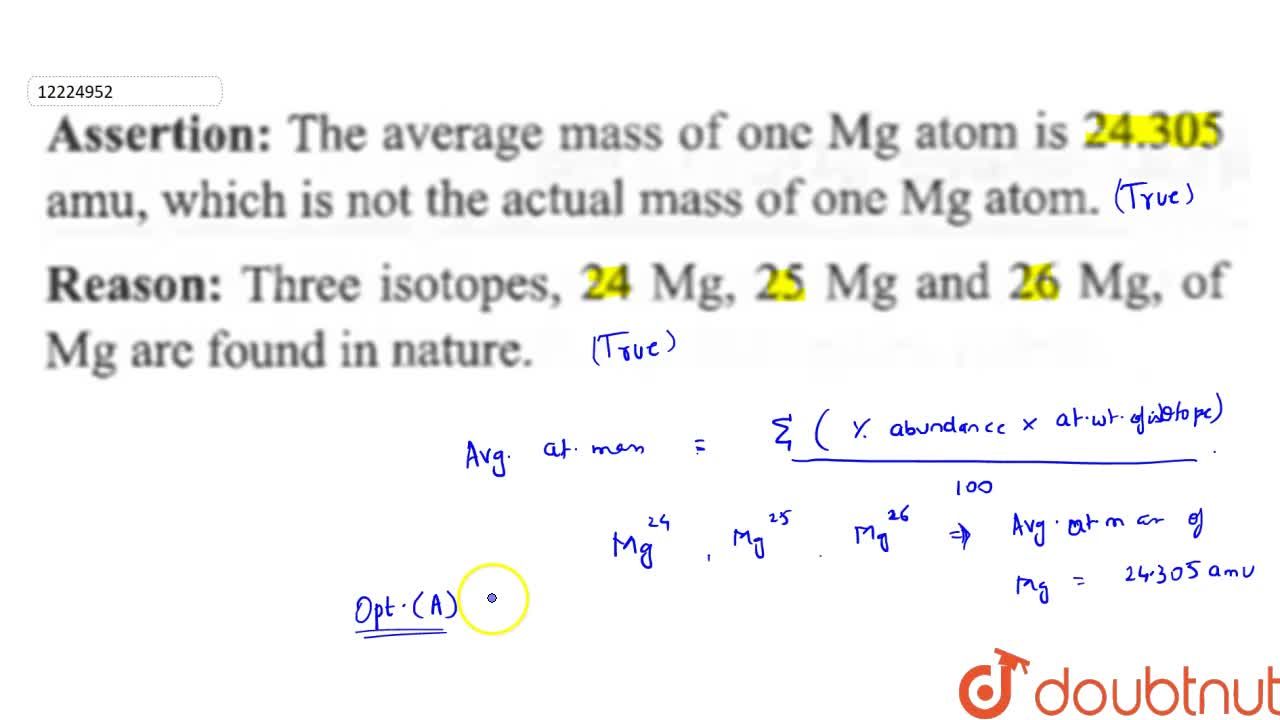In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their

SOLVED: Calculate the average atomic mass of magnesium using the following data for threc magnesium Isotopes Ksotope massl relativg abuudance Mg-24 23.985 0.7870 Mg-25 24.986 0.1013 Mg-26 25,983 0.1417 Calculatc the average

Average atomic mass of magnesium is 24.31 amu. This magnesium is composed of 79 mole % of .^(24)Mg and remaining 21 mol % of .^(25)Mg and .^(26)Mg. Calculate mole % of .^(26)Mg.

SOLVED: Magnesium has three naturally occurring isotopes with masses of 24, 25,26 a.m.u and natural abundances of 79%, 10% and 11%. Calculate the atomic mass of magnesium in U or Da; Sketch

2. Using the following data, calculate the average atomic mass of magnesium (give your answer to the - Brainly.com

Magnesium has two isotopes ^2412Mg and ^2612Mg . According to which isotopes of magnesium have different mass numbers.